Index
General principle of molecular imprinting
The technology of molecular imprinting
allows for the preparation of synthetic polymers with specific binding
sites for a target molecule. This can be achieved if the target is
present during the polymerization process, thus acting as a molecular
template. Monomers carrying certain functional groups are arranged around
the template through either noncovalent or covalent interactions. Following
polymerization with a high degree of cross-linking, the functional groups
are held in position by the polymer network. Subsequent removal of the
template by solvent extraction or chemical cleavage leaves cavities that
are complementary to the template in terms of size, shape and arrangement
of functional groups. These highly specific receptor sites are capable
of rebinding the target molecule with a high specificity, sometimes comparable
to that of antibodies. Molecularly imprinted polymers have therefore been
dubbed "antibody mimics". It has been shown that they can be substituted
for biological receptors in certain formats of immunoassays and biosensors.
They have also been used as stationary phases for affinity separations,
for the screening of combinatorial libraries, and as enzyme mimics in catalytic
applications.
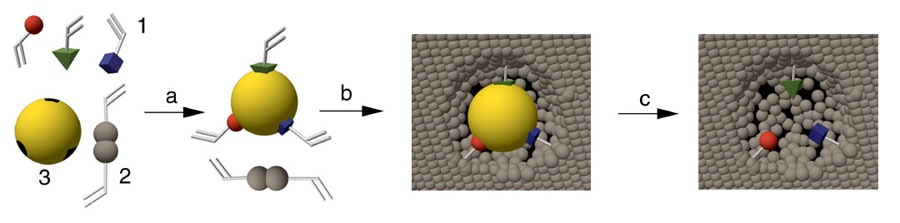
Schematic representation of the molecular imprinting process
1: Functional monomers, 2: cross-linker, 3: template molecule; a: assembly of the pre-polymerisation complex, b: polymerisation, c: extraction of the template liberating the binding site
Imprinting matrices
Organic polymers
Poly(meth)acrylates and polivinyl polymers
synthesised by radical polymerisation (the most
common)
Polyurethane
Polyaminophenyl boronate
Poly(phenylene diamine)
Poly(phenole)
Overoxidised polypyrrole
Sol-gels
Silica
Titanium dioxide
For a polymethacrylate MIPs specific for S-propranolol, synthesised by radical polymerisation, one needs the following ingredients :
| Ingredient |
Cross-linker |
Functional monomer(s) |
Template |
Polymerisation
initiator |
Porogenic solvent |
| Example |
Ethyleneglycol
dimethacrylate |
Methacrylic acid |
S-propranolol |
2,2’-Azobis-iso- butyronitrile (AIBN) |
Toluene |
| Structure |
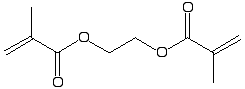
|
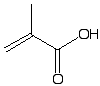
|

|
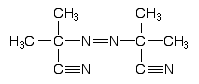
|
|
|
Typical
amount
(mmol) |
40 |
8 |
1 |
0.88 (1 mol-% of number
of polymerisable double bonds : here 88 mmol) |
1 ... 1.5 x volume of monomers |
Oxygen has to be removed by bubbling with nitrogen or argon for 3 min
on ice.
Polymerisation can be initiated by heating the mixture to
65 °C, or by UV irradiation at 350 nm.
The different synthesis and processing steps are shown below
:
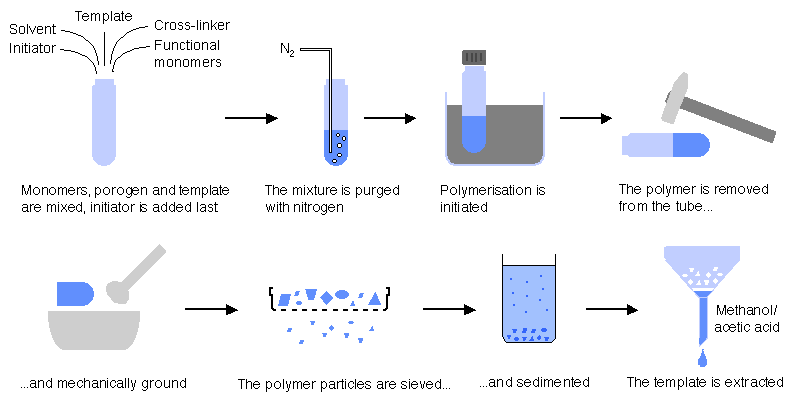
Characteristics of molecularly
imprinted polymers
Physical Stability
Resistant against mechanical stress, high pressures and elevated temperatures
Chemical Stability
Resistant against various organic solvents, against acids and bases depending on the material
Storage Endurance
typically > 1 year at room temperature without loss of performance
Imprint Memory
Repeated use >100 times without loss of memory
Binding Strength
< nM range (best sites)
Amino acidsImprinting of larger structures is more tricky :
Peptides
Steroids
Carbohydrates
Nucleotide bases
Others (pesticides, drugs etc.)
Metal ions
Proteins
Cells
Antibody / receptor binding site mimics
Immunoassays
Drug development / screening
Chemical sensors and biosensors
Tailor-made separation materials
Enantioseparation and separation of other closely related molecules (HPLC, CEC, TLC etc.)
Solid-phase extraction for sample preparation
Facilitated synthesis / Catalysis
Biomedical
Protecting groups
Equilibrium shifting
Catalytic polymers (enzyme mimics)
Slow release matrices
In-situ or extracorporeal removal of unwanted molecules
Our activities in the area
![]() Molecularly imprinted stationary
phases for separation
Molecularly imprinted stationary
phases for separation
(Enantioselective) membranes
Solid-phase extraction matrices
Capillary electrophoresis
![]() Sensors based on imprinted polymers
Sensors based on imprinted polymers
Electrochemical sensors
Infrared evanescence-wave sensors
Acoustic sensors (Quartz crystal microbalance)
![]() Immuno-type binding assays based
on imprinted polymers
Immuno-type binding assays based
on imprinted polymers
Radioassays
Fluorescence assays
Enzyme-linked assays (chemiluminescence and colorimetric)
High-throughput imaging formats
Flow injection capillary assays
Homogeneous assays
![]() Rational design and optimisation
of imprinted polymers
Rational design and optimisation
of imprinted polymers